Solving Kids’ Cancer Firsts
Solving Kids’ Cancer Firsts: A Decade of Breakthroughs & Leadership
Solving Kids’ Cancer is not just our name — it’s our mission. We target the toughest childhood cancers with the lowest survival rates because every kid deserves to grow up. Through global collaboration, we drive new treatments forward like combinations, sequences, and targeted cancer immunotherapy approaches. Our commitment is to wisely invest in innovative projects, making hope tangible and cures a reality for children and families everywhere. Here is a comprehensive overview of the impact we’ve made over the last decade — and it’s all thanks to you.
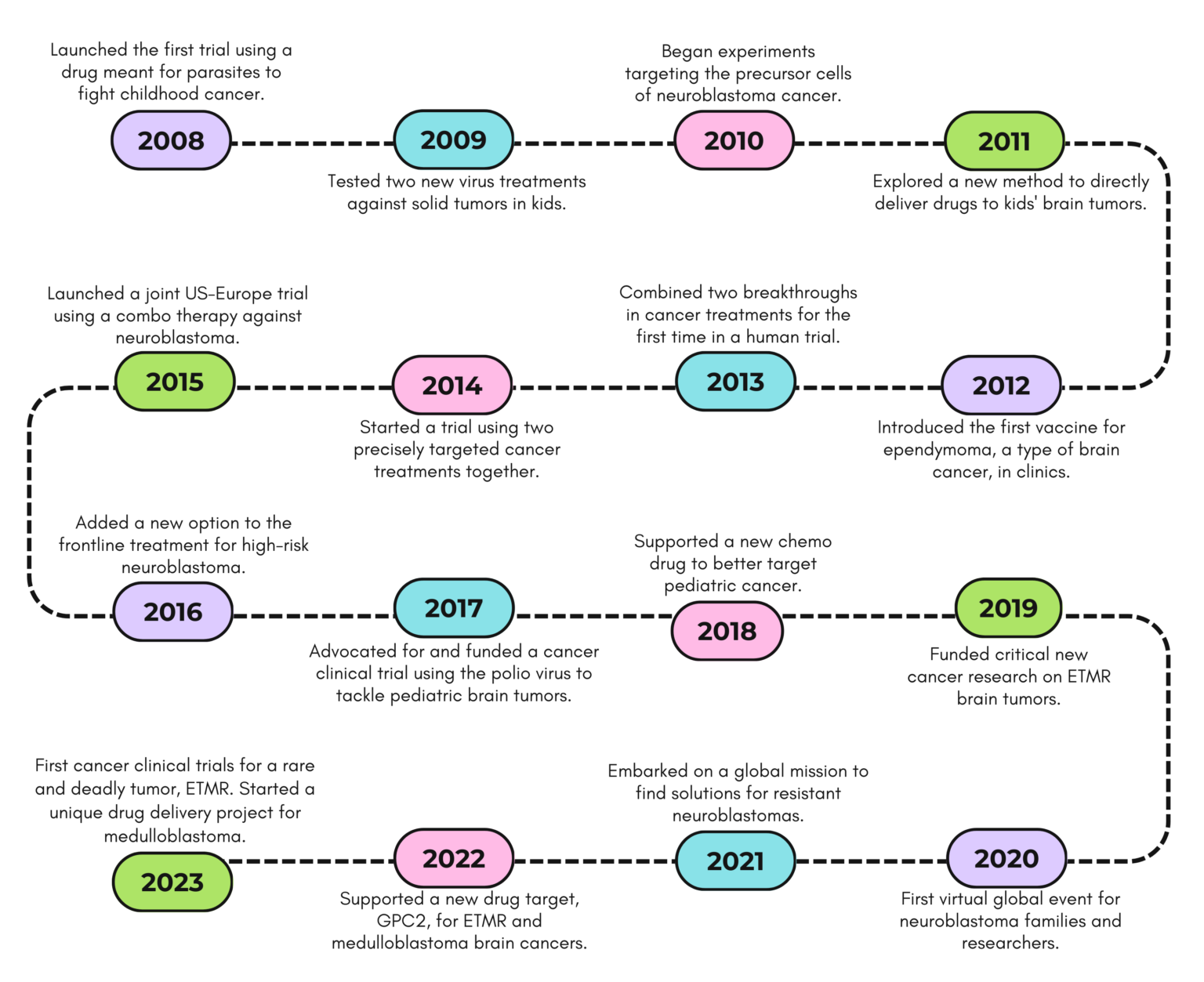
A Timeline of Innovation
For over ten years, Solving Kids’ Cancer has been at the forefront of cancer research and advocacy, marking each year with significant milestones that have shaped the landscape of pediatric oncology.
Making a Global Impact
Globally recognized. Locally impactful. Our presence resonates, ensuring the fight against childhood cancer never slows down.

• Patient Expertise: By integrating patient experiences and viewpoints, as well as knowledge of the cancer research landscape, we review and ensure that clinical trials are not only scientifically rigorous but also meet the real needs of children affected by cancer.
• Advocate, Pediatric and Adolescent Solid Tumor Steering Committee, National Cancer Institute (NCI), (2023-present)

• Collaboration in Drug Development: This collaborative effort with expert stakeholders including advocacy, industry, academia, and regulatory agencies focusing on speeding up the testing and approvals of novel drugs for patients with pediatric cancer.
• Member, Pediatric Drug Development (Fit for Filing), Accelerate Working Group 2, (2015-present)

• Quality Oversight: Our crucial advocacy informs Health Technology agencies such as the UK’s National Institute for Health and Care Excellence (NICE), influencing national drug approvals.
• National Institute for Health and Care Excellence (NICE), Drug Appraisals, (2015-present)

• International Coalitions: Working with like-minded charities in several countries we build collaborations to initiate and fund transatlantic cancer clinical trials, to ensure that better therapies are accessible to more children.
• Founding Board Member, Coalition Against Childhood Cancer (2012-present)

• Reviewing Pediatric Research: Serving on the NCI’s Central Review Board, we ensure that cancer clinical trials are conducted according to ethical regulations and that the risk/benefit ratio of therapies are appropriate for children battling cancer.
• Member, National Cancer Institute (NCI), Pediatric Central Review Board, (2015-present)

• Advocacy in Cancer Immunotherapy: Our involvement enables us to play a vital role in enhancing the effectiveness and accessibility of immunotherapies for patients worldwide, reflecting our dedication to pioneering transformative cancer treatments.
• Member, Society for Immunotherapy of Cancer (SITC), (2013-present)

• Advocacy at the FDA: We influence policy and decision-making processes, ensuring that patient needs and experiences are at the forefront of FDA approvals and breakthroughs in cancer treatment.
• FDA Patient Representative and FDA Oncologic Drugs Advisory Committee, (2010-present)
Empowering the Community With Knowledge
Our research lights the path forward, igniting hope and discovery in the pursuit of cures for cancer. Each publication we author contributes to a growing foundation of knowledge, helping to guide the global medical community toward groundbreaking, life-saving treatments:
- 2022 first paper on guidance for fit for filing data in academic sponsored cancer clinical trials:
- “The Critical Role of Academic Clinical Trials in Pediatric Cancer Drug Approvals: Design, Conduct, and Fit for Purpose Data for Positive Regulatory Decisions” Read more
- 2022 first advocate-bioethics authored paper on ethical issues:
- “A Systematic Literature Review to Identify Ethical, Legal, and Social Responsibilities of Nonprofit Organizations When Funding Clinical Trials in Pediatric Cancer” Read more
- 2022 first advocate-authored paper on parent perspectives on the use of stem cell transplant for high-risk neuroblastoma:
- “To Transplant, or Not to Transplant? That is the Question. A Patient Advocate Evaluation of Autologous Stem Cell Transplant in Neuroblastoma” Read more
- 2021 first advocate-authored white paper published in leading Oncology Journal:
- “When Innovation and Commercialization Collide: A Patient Advocate View in Neuroblastoma” Journal of Clinical Oncology. Read more
Leading and Collaborating for a Cure
Solving Kids’ Cancer has pioneered projects pushing the boundaries of what’s possible. Our funding and leadership have facilitated major cancer research and treatment advancements such as:
- ETMR Breakthrough Cancer Treatment: Starting the very first treatment plan for a very rare kind of childhood cancer.
- International Drug Cancer Clinical Trials: Partnering with a large team to test new medicines in the toughest neuroblastoma cases.
- New CAR T-cell Treatment: Creating a new type of therapy using the body’s own cells to fight specific brain cancers in kids.
- Studying Cancer Genes: Reviewing the genes of pediatric cancer to help doctors pick the best treatment for each child with neuroblastoma that comes back or doesn’t get better with standard treatment.
- Sharing Knowledge Worldwide: Building a global network that lets doctors and researchers around the world share what they know about neuroblastoma to help kids faster and more effectively.
- Fighting for Medicine for All: Advocating to make sure children everywhere can get the latest cancer-fighting medicines.
Every project advances us towards a cure for cancer, so children can look forward to living full, healthy lives.
Pioneering a United Front for Global Research
By uniting global experts, we steer the direction of cancer research, advocating for a better future for every child. Solving Kids’ Cancer has continued to earn a seat at the table among childhood cancer research experts, contributing to the direction of therapeutic development worldwide:
- Global Research Initiatives: Involvement in major research efforts that span countries and continents, looking to uncover the next big breakthrough in cancer treatment.
- Expert Conferences and Panels: Speaking at and participating in international conferences, sharing insights and learning from others in the field.
- Advocacy for Research Funding: Pushing for more investment in childhood cancer research, ensuring that these critical studies receive the support they need.
- Partnerships with Research Institutions: Partnering with top cancer research centers to accelerate the development of new therapies and get them to children.
- Policy Influence: Working with policymakers to make sure that the needs and challenges of pediatric cancer research are understood and addressed at the highest levels.
Together, these efforts are creating a strong network that is advancing the cause of childhood cancer treatment and research.
Here’s to Another Decade of Progress
Research into pediatric cancer requires time, financial resources, public interest, and unwavering dedication. Over the last decade, we’ve gained tremendous momentum, but there’s still much more to accomplish. That’s why your support is crucial — it fuels our ongoing progress, transforming lives, giving hope, and bringing us closer to a future where every child has the chance to grow up.



















































































